
Methanol, is made by using a catalyst to react carbon monoxide with hydrogen at high temperature and pressure. Assuming that 450.0 of CO and 825 of H2O are allowed to react, answer the following questions. (Hint: First write the balanced chemical equation for this reaction.)
Which reactant is in excess?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:10
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1


Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
Methanol, is made by using a catalyst to react carbon monoxide with hydrogen at high temperature and...
Questions


Mathematics, 30.05.2020 00:06


Mathematics, 30.05.2020 00:06




Mathematics, 30.05.2020 00:57





Mathematics, 30.05.2020 00:57



English, 30.05.2020 00:57




Mathematics, 30.05.2020 00:57

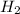 are allowed to react, answer the following questions. (Hint: First write the balanced chemical equation for this reaction.)
are allowed to react, answer the following questions. (Hint: First write the balanced chemical equation for this reaction.)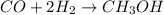
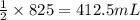 of carbon monoxide
of carbon monoxide

