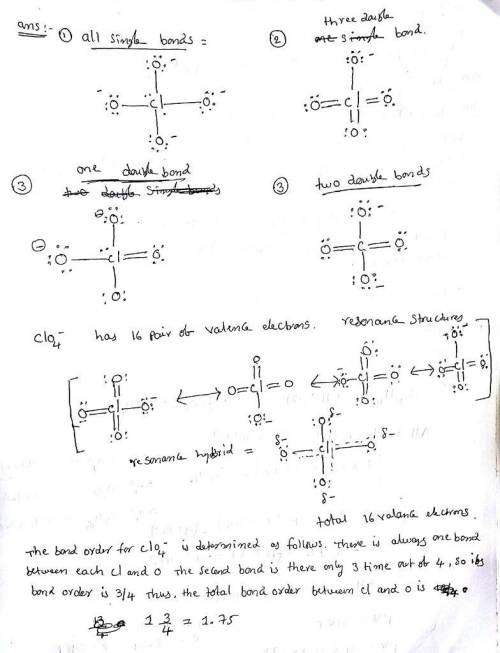
Erchlorates are powerful oxidizing agents used in fireworks, flares, and space shuttle booster rockets. Lewis structures for the perchlorate ion (ClO4−) can be drawn with all single bonds or with one, two, or three double bonds. Draw each of these possible resonance forms, including any nonbonding electrons. Include the values of any nonzero formal charges. Use formal charges to determine the most important resonance structure and calculate its average bond order.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

You know the right answer?
Erchlorates are powerful oxidizing agents used in fireworks, flares, and space shuttle booster rocke...
Questions

Biology, 24.09.2019 01:30



History, 24.09.2019 01:30

Mathematics, 24.09.2019 01:30

Mathematics, 24.09.2019 01:30






Geography, 24.09.2019 01:30


Computers and Technology, 24.09.2019 01:30


Mathematics, 24.09.2019 01:30



Advanced Placement (AP), 24.09.2019 01:30