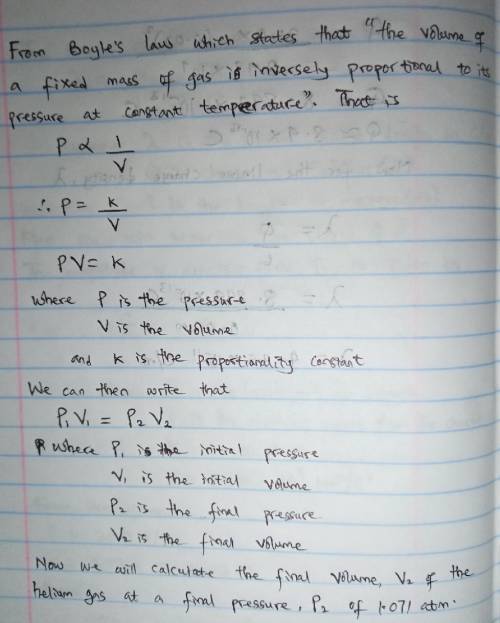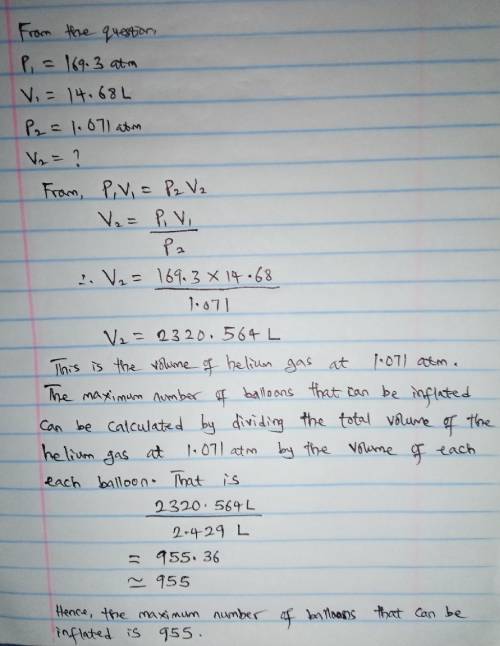Chemistry, 05.05.2020 22:21 mvazquez298
A cylinder containing 14.68 L of helium gas at a pressure of 169.3 atm is to be used to fill toy balloons to a pressure of 1.071 atm. Each inflated balloon has a volume of 2.429 L. What is the maximum number of balloons that can be inflated? Report your answer to 1 decimal place. (Remember that 14.68 L of helium at 1.071 atm will remain in the exhausted (empty) cylinder)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1
You know the right answer?
A cylinder containing 14.68 L of helium gas at a pressure of 169.3 atm is to be used to fill toy bal...
Questions


Mathematics, 04.04.2021 02:00

Physics, 04.04.2021 02:00

Mathematics, 04.04.2021 02:00


Computers and Technology, 04.04.2021 02:00


Mathematics, 04.04.2021 02:00

Mathematics, 04.04.2021 02:00








SAT, 04.04.2021 02:00


Mathematics, 04.04.2021 02:00

Mathematics, 04.04.2021 02:00