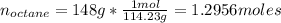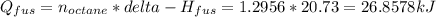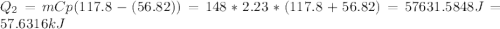Chemistry, 05.05.2020 19:12 alisonlebron15
Calculate the amount of heat needed to melt 148. g of solid octane (C8H18 ) and bring it to a temperature of 117.8 degrees c. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
An exothermic reaction is conducted in an insulated calorimeter filled with water. the calorimeter is then sealed so that there is no heat exchanged between the contents of the container and the surrounding air. which of the following statements is true about the reaction?
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 23.06.2019 03:00
Asample of sea water contains 6.28g of sodium chloride per litre of solution. how many milligrams of sodium chloride would be contained in 15.0ml of this solution?
Answers: 3

Chemistry, 23.06.2019 05:30
Find the midpoint of a segment with endpoints of 4-3i and -2+7i
Answers: 2
You know the right answer?
Calculate the amount of heat needed to melt 148. g of solid octane (C8H18 ) and bring it to a temper...
Questions



History, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Chemistry, 16.01.2020 07:31


Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31


Mathematics, 16.01.2020 07:31


Mathematics, 16.01.2020 07:31

Mathematics, 16.01.2020 07:31

Biology, 16.01.2020 07:31




Mathematics, 16.01.2020 07:31