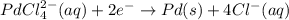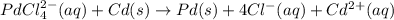Chemistry, 05.05.2020 18:31 cdradlet2001
A voltaic cell that uses the reaction PdCl42−(aq)+Cd(s) → Pd(s)+4Cl−(aq)+Cd2+(aq) has a measured standard cell potential of +1.03 V. You may want to reference (Pages 860 - 867) Section 20.4 while completing this problem. Part A Write the half-cell reaction at the cathode.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
A voltaic cell that uses the reaction PdCl42−(aq)+Cd(s) → Pd(s)+4Cl−(aq)+Cd2+(aq) has a measured sta...
Questions

Chemistry, 17.10.2020 21:01


Mathematics, 17.10.2020 21:01

Mathematics, 17.10.2020 21:01

Biology, 17.10.2020 21:01


English, 17.10.2020 21:01


Biology, 17.10.2020 21:01

English, 17.10.2020 21:01


Health, 17.10.2020 21:01

Mathematics, 17.10.2020 21:01


Mathematics, 17.10.2020 21:01