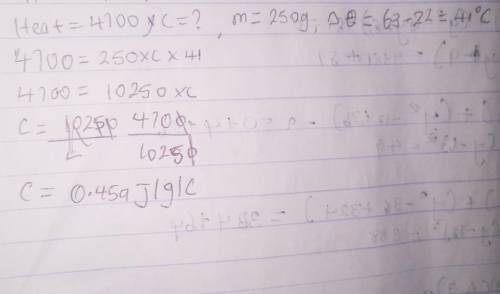Chemistry, 05.05.2020 17:18 mansnothot316
A student finds that 4700 J of heat are required to raise the temperature of 250.0 g of metal X from 22°C to 63°C. Based on the data in the table, what is the identity of metal X based upon finding the specific heat?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2


Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
A student finds that 4700 J of heat are required to raise the temperature of 250.0 g of metal X from...
Questions

Mathematics, 05.03.2021 05:30

Biology, 05.03.2021 05:30

Mathematics, 05.03.2021 05:30

Advanced Placement (AP), 05.03.2021 05:30

Mathematics, 05.03.2021 05:30

English, 05.03.2021 05:30

Biology, 05.03.2021 05:30

Biology, 05.03.2021 05:30

Health, 05.03.2021 05:30

Mathematics, 05.03.2021 05:30




English, 05.03.2021 05:30

Mathematics, 05.03.2021 05:30


History, 05.03.2021 05:30

Mathematics, 05.03.2021 05:30

History, 05.03.2021 05:30

English, 05.03.2021 05:30