
Chemistry, 05.05.2020 16:37 kaitlyn0123
100 mL of a strong acid is completely neutralized by 100 mL of a strong base. The observed products are salt and water. Upon further investigation, the solution has a pH > 7. How is this possible?
A) The concentration of acid is the same as the concentration of the base.
B) The concentration of acid is higher than the concentration of the base.
C) The concentration of base is higher than the concentration of the acid.
D) The water autoionizes to give [OH-] ions.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2


Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3
You know the right answer?
100 mL of a strong acid is completely neutralized by 100 mL of a strong base. The observed products...
Questions

Mathematics, 21.08.2019 18:00

English, 21.08.2019 18:00

Biology, 21.08.2019 18:00



Geography, 21.08.2019 18:00

Health, 21.08.2019 18:00

Mathematics, 21.08.2019 18:00


Mathematics, 21.08.2019 18:00

Physics, 21.08.2019 18:00


Mathematics, 21.08.2019 18:00

History, 21.08.2019 18:00

History, 21.08.2019 18:00





Mathematics, 21.08.2019 18:00

![pH = -log [H_3O^{+}]](/tpl/images/0640/3040/ad27d.png)
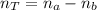 (1)
(1)

