
Chemistry, 05.05.2020 11:05 mckinneypaige8243
A 14.3 g sample of HF is dissolved into 250 mL of solution. The concentration of the solution is *
A. 2.86 M
B. 0.14 M
C. 7.1 M
D. 3.6 M

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1

Chemistry, 23.06.2019 05:30
Elizabeth has two separate samples of the same substance. sample is in the liquid state, and the other is in the solid state. the two samples most likely differ in which property?
Answers: 1
You know the right answer?
A 14.3 g sample of HF is dissolved into 250 mL of solution. The concentration of the solution is *
Questions

Mathematics, 03.02.2022 20:00

Computers and Technology, 03.02.2022 20:10

Chemistry, 03.02.2022 20:10


Biology, 03.02.2022 20:10



Social Studies, 03.02.2022 20:10

Health, 03.02.2022 20:10

Mathematics, 03.02.2022 20:10





Arts, 03.02.2022 20:10

Mathematics, 03.02.2022 20:20




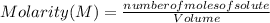
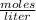 ).
).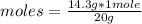
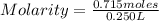
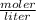 =2.86 M
=2.86 M

