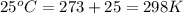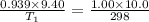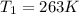Chemistry, 05.05.2020 09:57 alyssagonzales2021
A balloon of air occupies 10.0L(V2)at25.0°C(T2)and1.00atm(P2) . What temperature (T1) was it initially, if it occupied 9.40 L (V1) and was in a freezer with a pressure of 0.939 atm (P1)?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1

Chemistry, 23.06.2019 07:30
Achemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. the drug molecules bind the protein in a 1: 1 ratio to form a drug-protein complex. the protein concentration in aqueous solution at 25 ˚c is 1.74 x10-6 m . drug a is introduced into the protein solution at an initial concentration of 2.00 x10-6m. drug b is introduced into a separate, identical protein solution at an initial concentration of 2.00 x10-6m. at equilibrium, the drug a-protein solution has an a-protein complex concentration of 1.00 x10-6m, and the drug b solution has a b-protein complex concentration of 1.40 x10-6m.a. calculate the kc value for the a-protein binding reaction.b. calculate the kc value for the b-protein binding reaction.c. assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
Answers: 1

Chemistry, 23.06.2019 16:50
Consider the balanced equation below. pcl3+cl2-> pcl5 what is the mole ratio of pcl3 to pcl5
Answers: 1
You know the right answer?
A balloon of air occupies 10.0L(V2)at25.0°C(T2)and1.00atm(P2) . What temperature (T1) was it initial...
Questions

Biology, 17.11.2019 21:31





Mathematics, 17.11.2019 21:31






History, 17.11.2019 21:31


Business, 17.11.2019 21:31

Chemistry, 17.11.2019 21:31





Mathematics, 17.11.2019 21:31

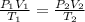
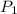 = initial pressure of gas = 0.939 atm
= initial pressure of gas = 0.939 atm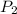 = final pressure of gas = 1.00 atm
= final pressure of gas = 1.00 atm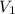 = initial volume of gas = 9.40 L
= initial volume of gas = 9.40 L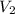 = final volume of gas = 10.0 L
= final volume of gas = 10.0 L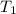 = initial temperature of gas = ?
= initial temperature of gas = ?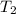 = final temperature of gas =
= final temperature of gas =