
Chemistry, 05.05.2020 09:07 mommytobe2019
Identify each part of this chemical equation, which describes the burning of methane and oxygen.
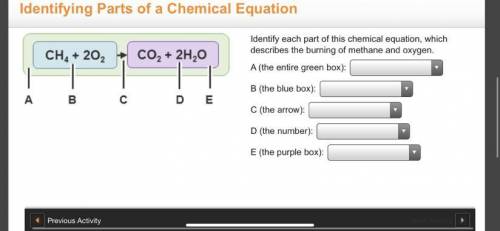

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following has wavelength longer than the wavelength of viable light? a) x rays b) gamma rays c) radios waves d) ultraviolet waves
Answers: 1

Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2
You know the right answer?
Identify each part of this chemical equation, which describes the burning of methane and oxygen.
Questions

Arts, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30


Mathematics, 14.12.2020 23:30




Mathematics, 14.12.2020 23:30

Social Studies, 14.12.2020 23:30

History, 14.12.2020 23:30


Physics, 14.12.2020 23:30






Computers and Technology, 14.12.2020 23:30

Mathematics, 14.12.2020 23:30

History, 14.12.2020 23:30



