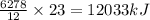Chemistry, 05.05.2020 00:44 Blakemiller2020
Using the following thermochemical equation, determine the amount of heat produced per kg of CO2 formed during the combustion of benzene (C6H6).
2 C6H6(l) + 15 O2(g) → 12 CO2(g) + 6 H2O(g) ΔH°rxn = -6278 kJ

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2


Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Using the following thermochemical equation, determine the amount of heat produced per kg of CO2 for...
Questions




Social Studies, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00


Health, 05.03.2021 14:00

Computers and Technology, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00


Mathematics, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00

Business, 05.03.2021 14:00

Biology, 05.03.2021 14:00

English, 05.03.2021 14:00

Social Studies, 05.03.2021 14:00


Biology, 05.03.2021 14:00

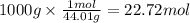
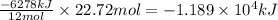
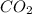 formed during the combustion of benzene
formed during the combustion of benzene 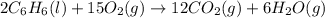
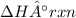 = -6278 kJ
= -6278 kJ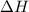 for the reaction comes out to be negative.
for the reaction comes out to be negative.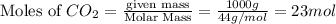 (1kg=1000g)
(1kg=1000g)