How many moles of H2O will be formed from the reaction of 80 g of
NaOH? *
H2SO4 + 2 NaOH...

Chemistry, 05.05.2020 03:43 mimireds8573
How many moles of H2O will be formed from the reaction of 80 g of
NaOH? *
H2SO4 + 2 NaOH = Na2SO4 + 2 H 0
0.5 moles
1 mole
2 moles
4 moles

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Write the complete balanced equation for the reaction between lead (iv) oxide (pbo2) and water (h2o).
Answers: 1

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 06:30
Over the last 90 years, scientists have added to the body of evidence supporting the big bang theory. what is the latest piece of evidence discovered in 2014?
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1
You know the right answer?
Questions



Mathematics, 12.02.2020 22:54



Mathematics, 12.02.2020 22:54

Social Studies, 12.02.2020 22:54

Mathematics, 12.02.2020 22:54


History, 12.02.2020 22:54


Mathematics, 12.02.2020 22:54






History, 12.02.2020 22:55


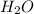 will be formed.
will be formed.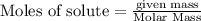
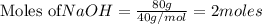
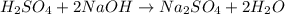
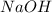 give = 2 moles of
give = 2 moles of 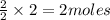 of
of 


