
Chemistry, 05.05.2020 16:44 willwhitlock803
A critical reaction in the production of energy in biological systems is the hydrolysis of adenosine triphosphate (ATP) to adenosine diphosphate (ADP) as described by ATP(aq) + H2O(l) ADP(aq) + HPO4-2(aq) for which ΔG°rxn = –30.5 kJ/mol at 37.0°C. Calculate the value of ΔGrxn in a biological cell in which [ATP] = 1.2 x10-2 M, [ADP] = 8.4 x10-3 M, and [HPO4-2] = 2.1x10-3 M.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 08:00
Match the vocabulary terms to their definitions. 1 . a long, chain-like set of molecules made up of repeating units joined end to end polymer 2 . a hard, brittle, heat- and corrosion-resistant material made by subjecting a nonmetallic mineral mixture to intense heat ceramic 3 . a plastic with low elongations that cannot be recycled thermoset 4 . a carbon fiber embedded in a polymer resin matrix thermoplastic 5 . a plastic with high elongations that can be recycled crystal 6 . a solid form resulting from the arrangement of atoms, ions, or molecules in definite geometric patterns composite
Answers: 1

Chemistry, 23.06.2019 10:00
How many moles are equal to 2.4×10^23 formula units of sodium chloride
Answers: 1
You know the right answer?
A critical reaction in the production of energy in biological systems is the hydrolysis of adenosine...
Questions

Health, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00

Social Studies, 23.10.2019 06:00





Social Studies, 23.10.2019 06:00


Spanish, 23.10.2019 06:00


Chemistry, 23.10.2019 06:00


Spanish, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00


History, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00

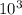 J/mol
J/mol ![\frac{[HPO4-2] x [ADP]}{ATP}](/tpl/images/0640/3718/8cebd.png)
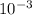 M
M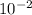 M
M![\frac{[2.1x10^{-3}] x [8.4x10^{-3}] }{[1.2 x 10^{-2}] }](/tpl/images/0640/3718/55eb3.png)


