
Chemistry, 05.05.2020 18:40 lillianneal
Substance ΔG°f(kJ/mol) M3O4(s) −8.80 M(s) 0 O2(g) 0 Consider the decomposition of a metal oxide to its elements, where M represents a generic metal. M3O4(s)↽−−⇀ 3M(s)+2O2(g) What is the standard change in Gibbs energy for the reaction, as written, in the forward direction? ΔG∘rxn= kJ/mol What is the equilibrium constant of this reaction, as written, in the forward direction at 298 K? K= What is the equilibrium pressure of O2(g) over M(s) at 298 K?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

You know the right answer?
Substance ΔG°f(kJ/mol) M3O4(s) −8.80 M(s) 0 O2(g) 0 Consider the decomposition of a metal oxide to i...
Questions

Mathematics, 18.09.2021 14:00



English, 18.09.2021 14:00

Mathematics, 18.09.2021 14:00

Mathematics, 18.09.2021 14:00






Social Studies, 18.09.2021 14:00

History, 18.09.2021 14:00


Mathematics, 18.09.2021 14:00


Chemistry, 18.09.2021 14:00

Health, 18.09.2021 14:00

World Languages, 18.09.2021 14:00

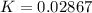
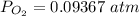
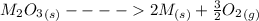
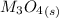 is
is 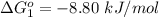
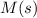 is
is 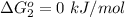
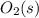 is
is 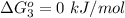
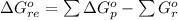
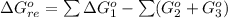
![\Delta G^o_{re} =[ (2 * 0) + (\frac{3}{2} * 0 )] - [1 * - 8.80]](/tpl/images/0641/3844/374c7.png)
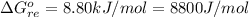

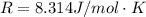
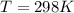
![K_p =[ P_{O_2}]^{\frac{3}{2} }](/tpl/images/0641/3844/66043.png)
![[ P_{O_2}]](/tpl/images/0641/3844/bc839.png) is the equilibrium pressure of oxygen
is the equilibrium pressure of oxygen 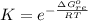
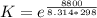
![0.02867 = [P_{O_2}]^{[\frac{3}{2} ]}](/tpl/images/0641/3844/d1a4b.png)
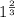
![P_{O_2} = [0.02867]^{\frac{2}{3} }](/tpl/images/0641/3844/dba69.png)


