
Chemistry, 06.05.2020 01:35 cbelew0001ouje4i
Which of the following item(s) explain the differences between the Ka values. Choose one or more: A. The oxidation state for oxygen in trifluoroacetate is more negative than the oxidation state for oxygen in acetate. B. The trifluoroacetate molecule has more resonance structures than the acetate molecule. C. The electron-withdrawing fluorine atoms pull electron density from the oxygen in trifluoroacetate. The negative charge is more stabilized in trifluoroacetate by this effect. D. The negative charge is on the more electronegative fluorine atom in trifluoroacetate.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:10
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1


Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
Which of the following item(s) explain the differences between the Ka values. Choose one or more: A....
Questions



Mathematics, 22.10.2020 23:01

Mathematics, 22.10.2020 23:01

Mathematics, 22.10.2020 23:01


English, 22.10.2020 23:01





Social Studies, 22.10.2020 23:01


English, 22.10.2020 23:01


Mathematics, 22.10.2020 23:01

Social Studies, 22.10.2020 23:01

Biology, 22.10.2020 23:01

Physics, 22.10.2020 23:01

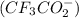 ,The acetate anion Two oxygen atoms.
,The acetate anion Two oxygen atoms.


