
Chemistry, 06.05.2020 06:44 blackchina71
If 20.5 g of chlorine is reacted with 20.5 g of sodium, which reactant is in excess?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 23.06.2019 07:00
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table?
Answers: 2
You know the right answer?
If 20.5 g of chlorine is reacted with 20.5 g of sodium, which reactant is in excess?...
Questions

Mathematics, 22.11.2019 07:31


Mathematics, 22.11.2019 07:31


Social Studies, 22.11.2019 07:31

Geography, 22.11.2019 07:31



English, 22.11.2019 07:31

World Languages, 22.11.2019 07:31


English, 22.11.2019 07:31

Mathematics, 22.11.2019 07:31


Spanish, 22.11.2019 07:31





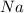 is in excess.
is in excess.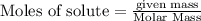
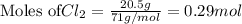

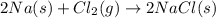
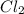 require 2 moles of
require 2 moles of 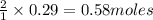 of
of 

