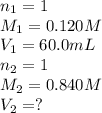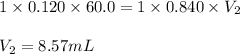Chemistry, 06.05.2020 20:11 vannybelly83
A volume of 60.0 mL of a 0.120 M HNO3 solution is titrated with 0.840 M KOH. Calculate the volume of KOH required to reach the equivalence point.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Which statement describes the appearance of a temperature-vs.-time graph? a horizontal line shows that the temperature increases at a constant rate over time. a vertical line shows that the temperature decreases at a constant rate over time. horizontal lines where the temperature is constant during phase changes connect upward-sloping lines where the temperature increases. horizontal lines where the temperature increases are connected by upward-sloping lines where the temperature is constant for each phase.
Answers: 1

Chemistry, 21.06.2019 16:50
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2

Chemistry, 21.06.2019 18:20
Which statement accurately describes the relationship between air pressure, air density, or altitude? as altitude increases, pressure increases.as altitude increases, air density increases.air pressure and density are lowest at sea level.denser air exerts more pressure than less dense air.
Answers: 2

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3
You know the right answer?
A volume of 60.0 mL of a 0.120 M HNO3 solution is titrated with 0.840 M KOH. Calculate the volume of...
Questions




Mathematics, 18.05.2021 23:30




History, 18.05.2021 23:30

English, 18.05.2021 23:30



Social Studies, 18.05.2021 23:30

Mathematics, 18.05.2021 23:30

Mathematics, 18.05.2021 23:30


Social Studies, 18.05.2021 23:30

Mathematics, 18.05.2021 23:30


English, 18.05.2021 23:30

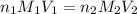
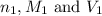 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 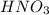
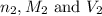 are the n-factor, molarity and volume of base which is KOH.
are the n-factor, molarity and volume of base which is KOH.