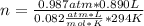Chemistry, 06.05.2020 21:19 manoli5874
How many miles of gas are contained in 890.0 mL at 21.0 C and 0.987 atm

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
How many miles of gas are contained in 890.0 mL at 21.0 C and 0.987 atm...
Questions







Arts, 29.06.2019 08:30

Biology, 29.06.2019 08:30

Social Studies, 29.06.2019 08:30

Social Studies, 29.06.2019 08:30


Chemistry, 29.06.2019 08:30




Biology, 29.06.2019 08:30



Mathematics, 29.06.2019 08:30

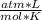 T= 21 °C= 294 °K
T= 21 °C= 294 °K