
Chemistry, 07.05.2020 00:58 juicemankinnie95
A 25.0 mL aliquot of 0.0430 0.0430 M EDTA EDTA was added to a 56.0 56.0 mL solution containing an unknown concentration of V 3 + V3+ . All of the V 3 + V3+ present in the solution formed a complex with EDTA EDTA , leaving an excess of EDTA EDTA in solution. This solution was back-titrated with a 0.0490 0.0490 M Ga 3 + Ga3+ solution until all of the EDTA EDTA reacted, requiring 13.0 13.0 mL of the Ga 3 + Ga3+ solution. What was the original concentration of the V 3 + V3+ solution?

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2


Chemistry, 23.06.2019 10:20
An engineer wishes to design a container that will hold 12.0 mol of ethane at a pressure no greater than 5.00x10*2 kpa and a temperature of 52.0 degrees celsius. what is the minimum volume the container can have?
Answers: 1
You know the right answer?
A 25.0 mL aliquot of 0.0430 0.0430 M EDTA EDTA was added to a 56.0 56.0 mL solution containing an un...
Questions

Mathematics, 02.11.2019 04:31

Social Studies, 02.11.2019 04:31



Mathematics, 02.11.2019 04:31




Social Studies, 02.11.2019 04:31



Physics, 02.11.2019 04:31

Biology, 02.11.2019 04:31



English, 02.11.2019 04:31


Biology, 02.11.2019 04:31


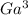 + and EDTA and
+ and EDTA and 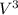 +
+
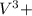 = 0.001075 - 0.00044 = 0.000675 moles
= 0.001075 - 0.00044 = 0.000675 moles


