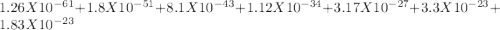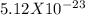Chemistry, 07.05.2020 02:07 UnusualKc2358
EDTA EDTA is a hexaprotic system with the p K a pKa values: p K a1 = 0.00 pKa1=0.00 , p K a2 = 1.50 pKa2=1.50 , p K a3 = 2.00 pKa3=2.00 , p K a4 = 2.69 pKa4=2.69 , p K a5 = 6.13 pKa5=6.13 , and p K a6 = 10.37 pKa6=10.37 . The distribution of the various protonated forms of EDTA will therefore vary with pH. For equilibrium calculations involving metal complexes with EDTA EDTA , it is convenient to calculate the fraction of EDTA EDTA that is in the completely unprotonated form, Y 4 − Y4− . This fraction is designated α Y 4 − αY4− . Calculate α Y 4 − αY4− at two pH values.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 23.06.2019 06:40
A250 g sample of water with an initial temperatureof 98.8 closes 6500 joules of heat. what is the finaltemperature of the water?
Answers: 1
You know the right answer?
EDTA EDTA is a hexaprotic system with the p K a pKa values: p K a1 = 0.00 pKa1=0.00 , p K a2 = 1.50...
Questions

Mathematics, 17.11.2019 08:31



Mathematics, 17.11.2019 08:31




Mathematics, 17.11.2019 08:31

Physics, 17.11.2019 08:31




Mathematics, 17.11.2019 08:31



World Languages, 17.11.2019 08:31



History, 17.11.2019 08:31

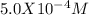
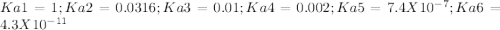
![alpha[Y^-4] = [H+]^6 + Ka1[H+]^5 + Ka1Ka2[H+]^4 + Ka1Ka2Ka3[H+]^3 + Ka1Ka2Ka3Ka4[H+]^2 + Ka1Ka2Ka3Ka4Ka5[H+] + Ka1Ka2Ka3Ka4Ka5Ka6](/tpl/images/0651/1171/e46f9.png)
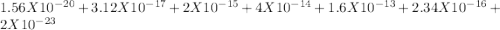
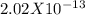
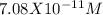
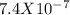 ; Ka6 =
; Ka6 = 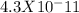
![alpha[Y^{-4}] = [H+]^6 + Ka1[H+]^5 + Ka1Ka2[H+]^4 + Ka1Ka2Ka3[H+]^3 + Ka1Ka2Ka3Ka4[H+]^2 + Ka1Ka2Ka3Ka4Ka5[H+] + Ka1Ka2Ka3Ka4Ka5Ka6](/tpl/images/0651/1171/ee319.png)