Chemistry, 07.05.2020 03:11 parrazm2022
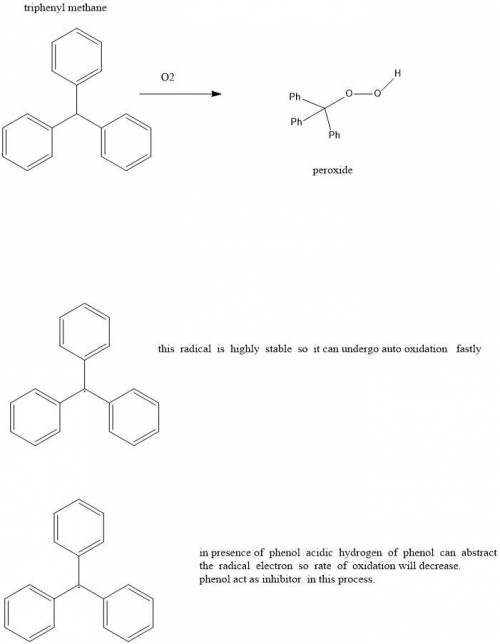
10.31 Triphenylmethane readily undergoes autooxidation to produce a hydroperoxide: c10s172 (a) Draw the expected hydroperoxide. (b) Explain why triphenylmethane is so susceptible to autooxidation. (c) In the presence of phenol (C6H5OH), triphenylmethane undergoes autooxidation at a much slower rate. Explain this observation.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 23.06.2019 01:00
Which substance—wood or silver—is the better thermal conductor? a thermal conductor is a material that requires very little heat energy to change its temperature. explain your answer.
Answers: 3
You know the right answer?
10.31 Triphenylmethane readily undergoes autooxidation to produce a hydroperoxide: c10s172 (a) Draw...
Questions

Physics, 02.10.2019 02:30

Computers and Technology, 02.10.2019 02:30

Computers and Technology, 02.10.2019 02:30


Health, 02.10.2019 02:30

Computers and Technology, 02.10.2019 02:30

Mathematics, 02.10.2019 02:30

History, 02.10.2019 02:30

English, 02.10.2019 02:30







Health, 02.10.2019 02:30


English, 02.10.2019 02:30

Mathematics, 02.10.2019 02:30