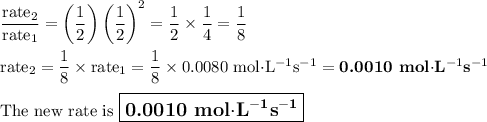The reaction is proceeding at a rate of 0.0080 Ms-1 in 50.0 mL of solution in a system with unknown concentrations of A and B. What is the rate immediately after the concentrations of all reactants are instantaneously lowered to 50% of their current value by adding an additional 50.0 mL of solvent?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1
You know the right answer?
The reaction is proceeding at a rate of 0.0080 Ms-1 in 50.0 mL of solution in a system with unknown...
Questions


Computers and Technology, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59



Health, 02.06.2020 05:59


English, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Biology, 02.06.2020 05:59


Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

Mathematics, 02.06.2020 05:59

![\dfrac{\text{rate}_{2}}{\text{rate}_{1}} = \dfrac{k_{2}\text{[A]}_2[\text{B]}_{2}^{2}}{k_{1}\text{[A]}_1[\text{B]}_{1}^{2}}= \left (\dfrac{\text{[A]}_{2}}{\text{[A]}_{1}}\right ) \left (\dfrac{\text{[B]}_{2}}{\text{[B]}_{1}}\right )^{2}](/tpl/images/0651/4550/63ca5.png)
![\dfrac{\text{[A]}_{2}}{\text{[A]}_{1}} = \dfrac{1}{2}\text{ and }\dfrac{\text{[B]}_{2}}{\text{[B]}_{1}}= \dfrac{1}{2}](/tpl/images/0651/4550/c7d0e.png)