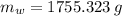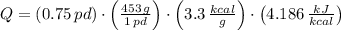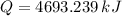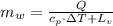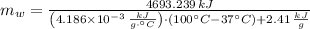Chemistry, 07.05.2020 05:12 marlesly87
The fuel value of hamburger is approximately 3.3 kcal/g. If a man eats 0.75 pounds of hamburger for lunch and none of the energy is stored in his body, estimate the amount of water that would have to be lost in perspiration to keep his body temperature constant. The heat of vaporization of water may be taken as 2.41 kJ/g.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 3

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2


Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1
You know the right answer?
The fuel value of hamburger is approximately 3.3 kcal/g. If a man eats 0.75 pounds of hamburger for...
Questions

History, 19.07.2019 15:50

History, 19.07.2019 15:50

Social Studies, 19.07.2019 15:50

Biology, 19.07.2019 15:50

Biology, 19.07.2019 15:50

Biology, 19.07.2019 15:50

Social Studies, 19.07.2019 15:50

Mathematics, 19.07.2019 15:50

Business, 19.07.2019 15:50

Biology, 19.07.2019 15:50

History, 19.07.2019 15:50

History, 19.07.2019 15:50




Mathematics, 19.07.2019 15:50




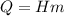
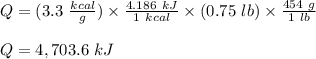
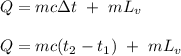
![Q = m[c(t_2 -t_1) \ + \ Lv]\\\\m = \frac{Q}{c(t_2 -t_1) \ + \ Lv} \\\\m = \frac{4,703.6 \times 10^3 }{4.186 (100-37) \ + \ 2.41 \times 10^3} \\\\m = 1,759.2 \ g\\\\m = 1.76 \ kg](/tpl/images/0651/6942/32919.png)