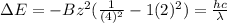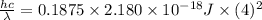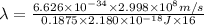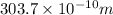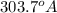Chemistry, 07.05.2020 05:15 granthazenp5e9mj
The energy of any one-electron species in its nth state (n = principal quantum number) is given by E = –BZ2 /n2 where Z is the charge on the nucleus and B is 18 2.180 10 J. a) Find the ionization energy of the Be3+ ion in its first excited state in kilojoules per mole. b) Find the wavelength of light given off from the Be3+ ion by electrons dropping from the fourth (n = 4) to the second (n = 2) energy levels.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1

Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2
You know the right answer?
The energy of any one-electron species in its nth state (n = principal quantum number) is given by E...
Questions



English, 12.06.2021 17:50



English, 12.06.2021 17:50


Mathematics, 12.06.2021 17:50

English, 12.06.2021 17:50

Mathematics, 12.06.2021 17:50


Mathematics, 12.06.2021 17:50

English, 12.06.2021 18:00

Social Studies, 12.06.2021 18:00




Chemistry, 12.06.2021 18:00


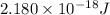
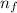 = 2; and
= 2; and 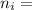 if it is ionized.
if it is ionized.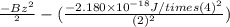
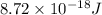
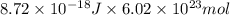
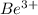 ion in its first excited state in kilojoules per mole is 5249 kJ/mol.
ion in its first excited state in kilojoules per mole is 5249 kJ/mol.