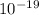Chemistry, 07.05.2020 06:06 reneecoutinho10
The addition of 5.0x10–3 total moles of Zn2+ to a 1.0 L solution of NaCN gives a solution of the complex ion [Zn(CN)4] 2– (Kf = 4.17x1019). What is the equilibrium concentration of uncomplexed Zn2+ ions if the concentration of cyanide ions at equilibrium is 0.50 M?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3
You know the right answer?
The addition of 5.0x10–3 total moles of Zn2+ to a 1.0 L solution of NaCN gives a solution of the com...
Questions



History, 01.09.2020 18:01


English, 01.09.2020 18:01

Mathematics, 01.09.2020 18:01


Spanish, 01.09.2020 18:01


Mathematics, 01.09.2020 18:01

Mathematics, 01.09.2020 18:01


Geography, 01.09.2020 18:01

Health, 01.09.2020 18:01

Mathematics, 01.09.2020 18:01

Mathematics, 01.09.2020 18:01

Mathematics, 01.09.2020 18:01



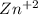 = 1.9 *
= 1.9 *