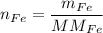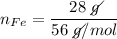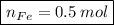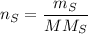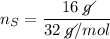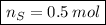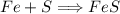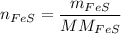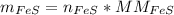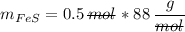Chemistry, 25.08.2019 06:00 malikbryant2002
In a chemical reaction, 28g of iron reacts with 16g of sulfur to produce of iron sulfide.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Activity two: just lemons, inc. production here's a one-batch sample of just lemons lemonade production. determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. hint: complete stoichiometry calculations for each ingredient to determine the theoretical yield. complete a limiting reactant-to-excess reactant calculation for both excess ingredients. water sugar lemon juice lemonade percent yield leftover ingredients 946.36 g 196.86 g 193.37 g 2050.25 g just lemons lemonade recipe equation: 2 water + sugar + lemon juice = 4 lemonade mole conversion factors: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g
Answers: 2

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

You know the right answer?
In a chemical reaction, 28g of iron reacts with 16g of sulfur to produce of iron sulfide....
Questions


Mathematics, 25.12.2021 14:00






Medicine, 25.12.2021 14:00

Biology, 25.12.2021 14:00

Health, 25.12.2021 14:00


History, 25.12.2021 14:00

Mathematics, 25.12.2021 14:00



Advanced Placement (AP), 25.12.2021 14:00

Mathematics, 25.12.2021 14:00


Physics, 25.12.2021 14:00

English, 25.12.2021 14:00