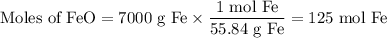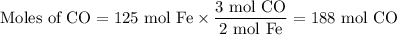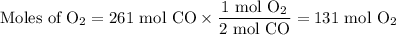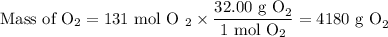Chemistry, 19.05.2020 02:17 mstrish71oteauw
Blast furnaces extra pure iron from the Iron(IIl)oxide in iron ore in a two step sequence. In the first step, carbon and oxygen react to form carbon monoxide:
2C(s)+O2(g) arrow 2CO(g)
In the second step, iron(lll) oxide and carbon monoxide react to form Iron and carbon dioxide:
Fe203(s) + 3CO(g) arrow 2Fe(s)+ 3CO2(g)
Suppose the yield of the first step is 71.% and the yield of the second step is 72.%. Calculate the mass of oxygen required to make 7.0 kg of iron.
Be sure your answer has a unit symbol, if needed, and is rounded to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 23.06.2019 02:00
The bone of a dinosaur and the imprint of a leaf are examples of which kind of fossils? a) index b) body c) amber d) trace
Answers: 1
You know the right answer?
Blast furnaces extra pure iron from the Iron(IIl)oxide in iron ore in a two step sequence. In the fi...
Questions



Mathematics, 23.11.2019 18:31



Biology, 23.11.2019 18:31

Health, 23.11.2019 18:31

History, 23.11.2019 18:31

Computers and Technology, 23.11.2019 18:31



Mathematics, 23.11.2019 18:31

Mathematics, 23.11.2019 18:31



English, 23.11.2019 18:31




Mathematics, 23.11.2019 18:31