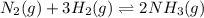Chemistry, 22.05.2020 03:05 mcmccann4317
Explain why it is necessary to balance chemical equations in terms of maintaining equilibrium. How can you tell when a chemical reaction has reached equilibrium? Give an example to help explain your answer.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Why does a plastic bottle that is sealed at a high altitude change it’s shape when taken to lower altitude
Answers: 2

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2
You know the right answer?
Explain why it is necessary to balance chemical equations in terms of maintaining equilibrium. How c...
Questions

Biology, 03.03.2021 23:20

English, 03.03.2021 23:20


History, 03.03.2021 23:20


Mathematics, 03.03.2021 23:20



Mathematics, 03.03.2021 23:20


Mathematics, 03.03.2021 23:20

Mathematics, 03.03.2021 23:20



Mathematics, 03.03.2021 23:20

Mathematics, 03.03.2021 23:20


Mathematics, 03.03.2021 23:20