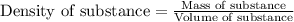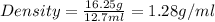Chemistry, 23.05.2020 04:03 ramjand7853
A graduated cylinder has 33.5 mL of water. A piece of metal is added to the water and the new volume is now 46.2 mL. If the metal has a mass of 16.25 g, what is its density

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2
You know the right answer?
A graduated cylinder has 33.5 mL of water. A piece of metal is added to the water and the new volume...
Questions



Mathematics, 22.10.2019 05:20




Mathematics, 22.10.2019 05:20


Mathematics, 22.10.2019 05:30

Mathematics, 22.10.2019 05:30





Computers and Technology, 22.10.2019 05:30

Mathematics, 22.10.2019 05:30

Mathematics, 22.10.2019 05:30



Chemistry, 22.10.2019 05:30