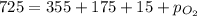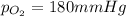Chemistry, 24.05.2020 07:57 meiaalvizo2005
A mixture of gases at a total pressure of 725 mmHg contains nitrogen, carbon dioxide, helium, and oxygen. The partial pressure of carbon dioxide is 175 mmHg, the partial pressure of nitrogen is 355 mmHg, and the partial pressure of helium is 15 mmHg. What is the partial pressure of oxygen?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
A mixture of gases at a total pressure of 725 mmHg contains nitrogen, carbon dioxide, helium, and ox...
Questions



Geography, 28.06.2019 07:30




Computers and Technology, 28.06.2019 07:30

English, 28.06.2019 07:30

History, 28.06.2019 07:30


Mathematics, 28.06.2019 07:30




Mathematics, 28.06.2019 07:30





Advanced Placement (AP), 28.06.2019 07:30

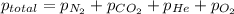
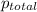 =total pressure of gases = 725 mm Hg
=total pressure of gases = 725 mm Hg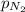 = partial pressure of nitrogen = 355 mm Hg
= partial pressure of nitrogen = 355 mm Hg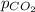 = partial pressure of carbon dioxide = 175 mm Hg
= partial pressure of carbon dioxide = 175 mm Hg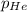 = partial pressure of helium = 15 mm Hg
= partial pressure of helium = 15 mm Hg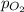 = partial pressure of oxygen = ?
= partial pressure of oxygen = ?