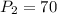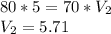Chemistry, 26.05.2020 01:59 hjeffrey168
A gas at 80 kPa occupies a volume of 5 mL. What volume will the gas occupy at 70 kPa

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Asap! ! who wants to me with my assignment : ) (especially with how i’m gonna draw out the example) you have explored some interesting, informative, and amusing examples of models. now it's time to get creative and make your own model. here is the requirement checklist for your model: ✔ model types can include drawings, diagrams, physical models, virtual simulations, or videos. ✔ models must be created by you, not something selected from an online or outside source. ✔ submit a presentation, picture, video, or screenshot of your model. ✔ submit a one-paragraph summary describing the topic you chose, your model, what it represents, how you made it, and the specific science involved. it is important that you are using science terminology and are accurate. now that you know how to create and submit your model, you will need to choose a topic for your model. choose one of the three topics listed below. select each topic for an overview. -conservation of mass -atomic theory -thermal energy
Answers: 2

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 11:00
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow.part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer.o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part.h2o is a stronger acid than oh–, so the equilibrium lies to the right.h2o is a weaker acid than oh–, so the equilibrium lies to the left.h2o is a stronger acid than oh–, so the equilibrium lies to the left.h2o is a weaker acid than oh–, so the equilibrium lies to the right.part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer.ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-(aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left.part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer.no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part.hno2 is a stronger acid than h2o, so the equilibrium lies to the right.hno2 is a weaker acid than h2o, so the equilibrium lies to the left.hno2 is a stronger acid than h2o, so the equilibrium lies to the left.hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1
You know the right answer?
A gas at 80 kPa occupies a volume of 5 mL. What volume will the gas occupy at 70 kPa...
Questions



World Languages, 14.05.2021 14:00



Biology, 14.05.2021 14:00


Mathematics, 14.05.2021 14:00

English, 14.05.2021 14:00

Mathematics, 14.05.2021 14:00




Mathematics, 14.05.2021 14:00

Geography, 14.05.2021 14:00

History, 14.05.2021 14:00


Chemistry, 14.05.2021 14:00

Mathematics, 14.05.2021 14:00

Mathematics, 14.05.2021 14:00

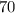 kPa
kPa 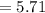
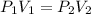
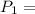 initial pressure of the gas
initial pressure of the gas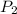 = final pressure of the gas
= final pressure of the gas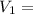 Initial volume of the gas
Initial volume of the gas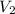 = the final volume of the gas
= the final volume of the gas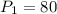 kPa
kPa 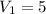 mL
mL