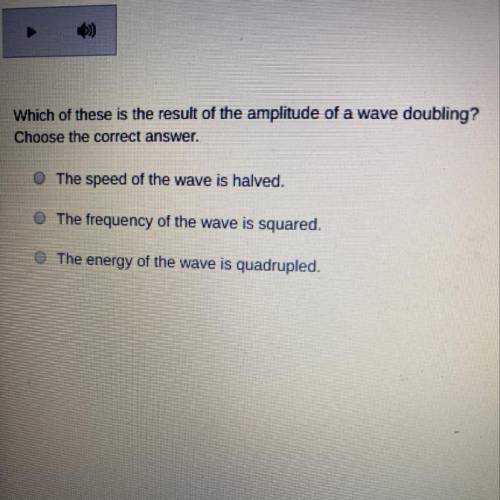
Can someone help please.
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:30
What is the molecular formula of a hydrocarbon with m+ = 166? (write the formula with no subscripts, e.g. c4h10.) what is the sum of rings and double bonds in this compound?
Answers: 1

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
Questions

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

World Languages, 11.09.2020 16:01

Social Studies, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

English, 11.09.2020 16:01

Physics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Physics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01