What is the mass in grams of 1.50 X 1023 molecules of NaOH?
a. 10.0 g
b. 20.0 g
...

Chemistry, 29.05.2020 05:57 fruitbutt77
What is the mass in grams of 1.50 X 1023 molecules of NaOH?
a. 10.0 g
b. 20.0 g
c. 30.0 g
d. 40.0 g
e. 60.0 g

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1


Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1
You know the right answer?
Questions


Mathematics, 22.05.2021 20:30





Mathematics, 22.05.2021 20:30

Mathematics, 22.05.2021 20:30



Mathematics, 22.05.2021 20:30


Physics, 22.05.2021 20:30



Social Studies, 22.05.2021 20:30

Mathematics, 22.05.2021 20:30


Mathematics, 22.05.2021 20:30

Mathematics, 22.05.2021 20:30

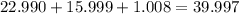 . But before we do anything with this number we must first convert
. But before we do anything with this number we must first convert  molecules to moles. We can do this by dividing by Avogadro's number or
molecules to moles. We can do this by dividing by Avogadro's number or 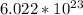 (
( 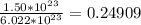 moles). We then convert moles to grams by multiplying by the molar mass of NaOH, (
moles). We then convert moles to grams by multiplying by the molar mass of NaOH, (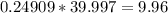 grams). This is almost exactly 10 and the margin of error is likely due to the original numbers not being precise enough.
grams). This is almost exactly 10 and the margin of error is likely due to the original numbers not being precise enough. 

