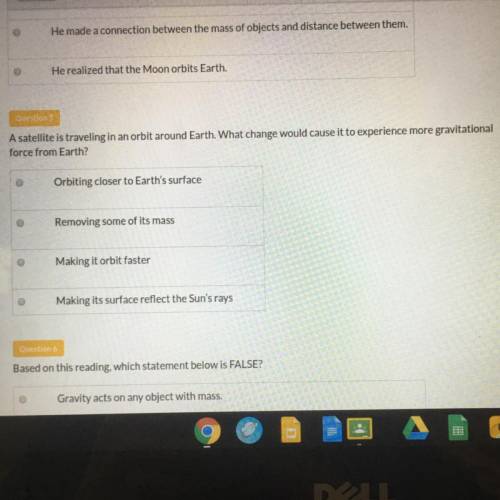
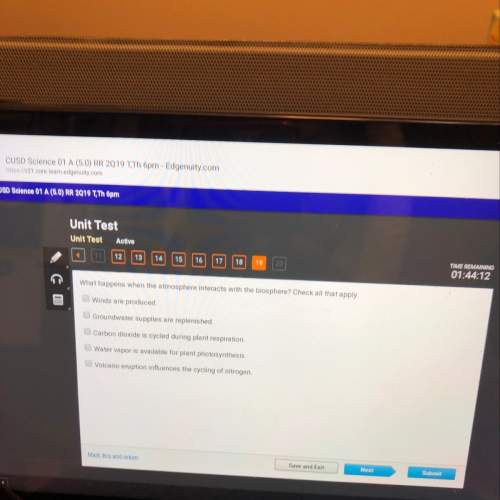
Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2
You know the right answer?
HELPPP TODAY IS THE LAST DAY OF SCHOOL
...
...
Questions

Mathematics, 25.11.2021 14:10

Mathematics, 25.11.2021 14:10



SAT, 25.11.2021 14:10

English, 25.11.2021 14:10


Mathematics, 25.11.2021 14:10

Social Studies, 25.11.2021 14:10


Chemistry, 25.11.2021 14:10




History, 25.11.2021 14:10




Medicine, 25.11.2021 14:10