
Chemistry, 30.05.2020 06:59 jackrider6563
10) Intravenous saline solutions have a concentration of 1.5 M NaCl. How many liters of saline can be prepared from 50.0 g of NaCl?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 15:30
Plz me ! 1 which of earths spheres contains most of its mass? a atmosphere b hydrosphere c geosphere* d biosphere 2 erosion and weathering are examples of which types of forces? a constructive forces b destructive forces* c gravitational forces d inertia-related forces 3 which of the following statements about earths atmosphere is true? a earths atmosphere contains 78% water vapor which is essentail to life b earths atmosphere contains 21% oxygen c earths atmosphere contains carbon dioxide which all life forms require d earths atmosphere allows radiation from the sun to pass through it and warm earths surface* 4 the strenght of the force of gravity between two objects is determined by which of the following factors? select all that apply a the messes of the objects* b the distance between the objects* c the volumes of the objects d the surface area of the objects 5 earth and moon are kept in there respective orbits due to the influence of a inertia b gravity c gravity and inertia* d neither gravity or inertia if you answer all questions right i will give
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
10) Intravenous saline solutions have a concentration of 1.5 M NaCl. How many liters of saline can b...
Questions



History, 19.10.2019 06:30




History, 19.10.2019 06:30



History, 19.10.2019 06:30

History, 19.10.2019 06:30


Biology, 19.10.2019 06:30

English, 19.10.2019 06:30

Mathematics, 19.10.2019 06:30

History, 19.10.2019 06:30

Mathematics, 19.10.2019 06:30

Chemistry, 19.10.2019 06:30

English, 19.10.2019 06:30

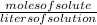 or Moles = molarity × litres
or Moles = molarity × litres

