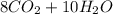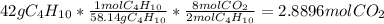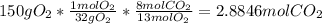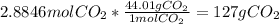Chemistry, 30.05.2020 05:00 hinokayleen5053
Gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water . Suppose 42. g of butane is mixed with 150. g of oxygen. Calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3


Chemistry, 22.06.2019 05:00
Type the letter that represents the correct location for each particle type below.
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1
You know the right answer?
Gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water ....
Questions

Health, 26.09.2019 02:00

Biology, 26.09.2019 02:00

Health, 26.09.2019 02:00

Physics, 26.09.2019 02:00

Mathematics, 26.09.2019 02:00


Health, 26.09.2019 02:00



Mathematics, 26.09.2019 02:00



Computers and Technology, 26.09.2019 02:00


Mathematics, 26.09.2019 02:00



Spanish, 26.09.2019 02:00


Social Studies, 26.09.2019 02:00

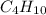 , oxygen is
, oxygen is 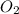 , carbon dioxide is
, carbon dioxide is 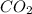 , and water is
, and water is 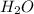 . The reactants are butane and oxygen and the products are carbon dioxide and water. So we write:
. The reactants are butane and oxygen and the products are carbon dioxide and water. So we write: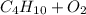 ⇒
⇒ 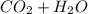
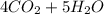
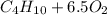 ⇒
⇒ 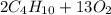 ⇒
⇒