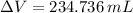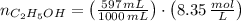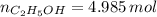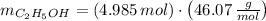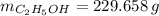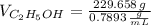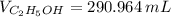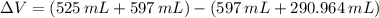A solution is prepared by mixing 525 mL of ethanol with 597 mL of water. The molarity of ethanol in the resulting solution is 8.35 M. The density of ethanol at this temperature is 0.7893 g/mL. Calculate the difference in volume between the total volume of water and ethanol that were mixed to prepare the solution and the actual volume of the solution. g

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 03:00
How does a hydroelectric power plant converts energy into energy.
Answers: 1

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

You know the right answer?
A solution is prepared by mixing 525 mL of ethanol with 597 mL of water. The molarity of ethanol in...
Questions

Mathematics, 08.12.2021 01:00

Mathematics, 08.12.2021 01:00


English, 08.12.2021 01:00


Chemistry, 08.12.2021 01:00

English, 08.12.2021 01:00

SAT, 08.12.2021 01:00




Mathematics, 08.12.2021 01:00

Mathematics, 08.12.2021 01:00

Mathematics, 08.12.2021 01:00