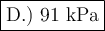Chemistry, 31.05.2020 05:00 jenadkrson62
There are 0.55 moles of carbon dioxide gas in a 15.0 L container. This container is at a temperature of 300 K. What is the pressure of the gas inside the container? Use 8.31 L*kPa/mol*K for the gas constant.
A.)760 mm Hg\
B.) 271 kPa
C.) 2 atm
D.) 91.4 kPa

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have weak covalent bonds. which of the following is most likely a property of this substance? a. high ph b. high conductivity c. low melting point d. low flammability
Answers: 3

Chemistry, 23.06.2019 10:00
State the effect on the concentration of the clo- ion when there is a decrease in the concentration of the oh- ion
Answers: 1
You know the right answer?
There are 0.55 moles of carbon dioxide gas in a 15.0 L container. This container is at a temperature...
Questions


Health, 10.10.2019 06:10

History, 10.10.2019 06:10


Mathematics, 10.10.2019 06:10

History, 10.10.2019 06:10

Biology, 10.10.2019 06:10

Mathematics, 10.10.2019 06:10

Chemistry, 10.10.2019 06:10






Biology, 10.10.2019 06:10





Mathematics, 10.10.2019 06:10