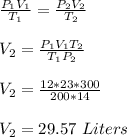Chemistry, 01.06.2020 12:57 potato3999
If I initially have a gas at a pressure of 12 atm, a volume of 23 Liters and a temperature of 200K, and then I raise the pressure to 14 atm and increase the temperature to 300K, what is the new volume?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3


Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
If I initially have a gas at a pressure of 12 atm, a volume of 23 Liters and a temperature of 200K,...
Questions


English, 04.02.2020 09:48

Mathematics, 04.02.2020 09:48

Computers and Technology, 04.02.2020 09:48



Mathematics, 04.02.2020 09:48



Mathematics, 04.02.2020 09:48



History, 04.02.2020 09:48

Chemistry, 04.02.2020 09:48