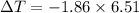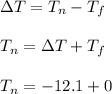What is the freezing point of a solution of 210.0 g of glycerol, formula C3H8O3, dissolved in 350. g of water? Careful. First get molar mass and use molar mass to determine molality concentration. Then use freeze pt. depression formula to determine the change in freezing pt. Then determine the new freeze point. The freezing point depression constant for water is Kf= -1.86 oCelcius/molal. Report your answer rounded to 1 decimal point and do not include units.

Answers: 2


Another question on Chemistry



Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1
You know the right answer?
What is the freezing point of a solution of 210.0 g of glycerol, formula C3H8O3, dissolved in 350. g...
Questions








Physics, 30.04.2021 20:50

Mathematics, 30.04.2021 20:50


Mathematics, 30.04.2021 20:50


Chemistry, 30.04.2021 20:50

Mathematics, 30.04.2021 20:50

Mathematics, 30.04.2021 20:50

Arts, 30.04.2021 20:50

Mathematics, 30.04.2021 21:00


Computers and Technology, 30.04.2021 21:00

History, 30.04.2021 21:00

 Freezing point depression constant for water, Kf = 0.512 °C/m
Freezing point depression constant for water, Kf = 0.512 °C/m
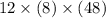
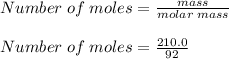
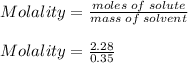
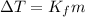
 is the change in temperature.
Kf is the molal freezing point constant.
m is the molality of solution.
is the change in temperature.
Kf is the molal freezing point constant.
m is the molality of solution.