N2(g) + 3H2(g) <-> 2NH3(g) + energy

Chemistry, 02.06.2020 19:58 roxymiller3942
Given the equation for a system at equilibrium:
N2(g) + 3H2(g) <-> 2NH3(g) + energy
If only the concentration of N2(g) is increased, the concentration of
1. NH3(g) increases
2. NH3(g) remains the same
3. H2(g) increases
4. H2(g) remains the same

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1

Chemistry, 23.06.2019 01:30
In which phase of mitosis do the spindle fibers pull the chromosomes apart to opposite sides of the cell ?
Answers: 1

Chemistry, 23.06.2019 11:20
Try to reduce the amount of leftover ingredients by changing the amount of one, two, or all three starting ingredients. show your stoichiometric calculations below. water 946.36 g sugar 196.86 g lemon juice193.37 g
Answers: 2

Chemistry, 23.06.2019 13:30
Which of the following is true regarding chemical and nuclear reactions?
Answers: 1
You know the right answer?
Given the equation for a system at equilibrium:
N2(g) + 3H2(g) <-> 2NH3(g) + energy
N2(g) + 3H2(g) <-> 2NH3(g) + energy
Questions








Computers and Technology, 09.08.2019 22:30









Computers and Technology, 09.08.2019 22:30



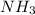 increases.
increases.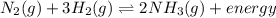
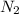 is increased , according to the Le-Chatlier's principle, the reaction has to shift to right or forward direction. In order to do that the concentration of products has to increase.
is increased , according to the Le-Chatlier's principle, the reaction has to shift to right or forward direction. In order to do that the concentration of products has to increase.

