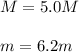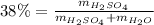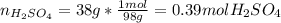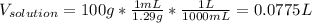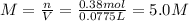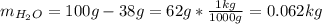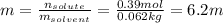Chemistry, 05.06.2020 08:59 msjsnell29
Automobile battery acid is 38% H2SO4 and has a destiny of 1.29g/ml. Calculate the molality and the molarity of this solution.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 03:00
Which of the dna typing techniques do you think you would choose if you had to analyze a dna sample? why?
Answers: 1

Chemistry, 22.06.2019 06:30
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3
You know the right answer?
Automobile battery acid is 38% H2SO4 and has a destiny of 1.29g/ml. Calculate the molality and the m...
Questions



Mathematics, 26.01.2021 14:00

Physics, 26.01.2021 14:00



History, 26.01.2021 14:00


World Languages, 26.01.2021 14:00

Biology, 26.01.2021 14:00

Biology, 26.01.2021 14:00


Health, 26.01.2021 14:00

Mathematics, 26.01.2021 14:00

Mathematics, 26.01.2021 14:00


Biology, 26.01.2021 14:00