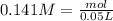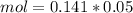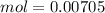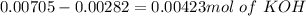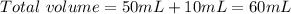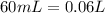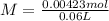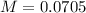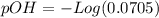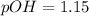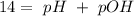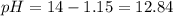Chemistry, 06.06.2020 03:59 genyjoannerubiera
A 50.0 mL solution of 0.141 M KOH is titrated with 0.282 M HCl . Calculate the pH of the solution after the addition of each of the given amounts of HCl .

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 09:20
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1


Chemistry, 23.06.2019 11:50
How many moles of an ideal gas would occupy a 25.0 liter container when the temperature is 295 k and the pressure is 0.850 atm?
Answers: 2
You know the right answer?
A 50.0 mL solution of 0.141 M KOH is titrated with 0.282 M HCl . Calculate the pH of the solution af...
Questions











Mathematics, 01.08.2019 09:00




Mathematics, 01.08.2019 09:00


History, 01.08.2019 09:00

Mathematics, 01.08.2019 09:00

Biology, 01.08.2019 09:00

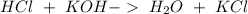
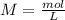
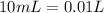 and we have the concentration of the HCl
and we have the concentration of the HCl 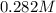 , when we plug the values into the equation we got:
, when we plug the values into the equation we got: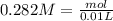
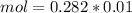
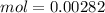
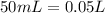 and
and 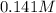 ).
).