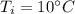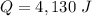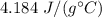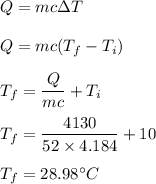Chemistry, 06.06.2020 04:00 zaniyastubbs9
A 52-gram sample of water that has an initial temperature of 10.0 °C absorbs 4,130 joules. If the specific heat of water is 4.184 J/(g °C), what is the final temperature of the water?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 19:20
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3
You know the right answer?
A 52-gram sample of water that has an initial temperature of 10.0 °C absorbs 4,130 joules. If the sp...
Questions

Biology, 02.07.2019 06:50



Mathematics, 02.07.2019 07:00


Mathematics, 02.07.2019 07:00


Geography, 02.07.2019 07:00

Mathematics, 02.07.2019 07:00

History, 02.07.2019 07:00


Mathematics, 02.07.2019 07:00


History, 02.07.2019 07:00

Mathematics, 02.07.2019 07:00


English, 02.07.2019 07:00



History, 02.07.2019 07:00