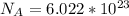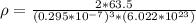Chemistry, 06.06.2020 21:01 gummybear0
Copper is a solid phase of copper still unknown to science. The only difference between it and ordinary copper is that Copper forms a crystal with a bcc unit cell and a lattice constant . Calculate the density of Copper . Round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2


You know the right answer?
Copper is a solid phase of copper still unknown to science. The only difference between it and ordin...
Questions



Arts, 14.05.2021 01:00

History, 14.05.2021 01:00



Mathematics, 14.05.2021 01:00



Mathematics, 14.05.2021 01:00

Mathematics, 14.05.2021 01:00

Spanish, 14.05.2021 01:00


Chemistry, 14.05.2021 01:00


Mathematics, 14.05.2021 01:00




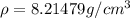
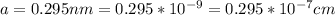
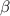 ) is mathematically represented as
) is mathematically represented as 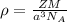
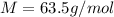
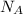 is the Avogadro's constant with a value of
is the Avogadro's constant with a value of