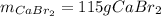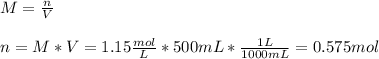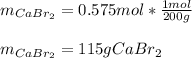Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1


Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
Determine the mass of CaBr2 needed to create a 500. ml solution with a concentration of 1.15 M....
Questions

English, 05.10.2019 16:30


Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30


Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30


Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30





Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30

Mathematics, 05.10.2019 16:30