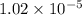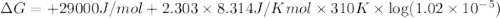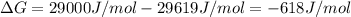Chemistry, 07.06.2020 05:00 21ghostrider21
The standard free energy change in physiological conditions (G') for the reaction catalyzed by malate dehydrogenase in the citric acid cycle malate + NAD+ oxaloacetate + NADH + H+ is ~+29 kJmol-1 Calculate the actual G' at 37C if Keq' is 1.02 × 10-5

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Complete the following reactions using word and balanced equations including states. dilute phosphoric acid is added with a calcium hydroxide solution.
Answers: 1

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
The standard free energy change in physiological conditions (G') for the reaction catalyzed by malat...
Questions


History, 02.04.2021 07:30

Mathematics, 02.04.2021 07:30



Mathematics, 02.04.2021 07:30

Mathematics, 02.04.2021 07:30

History, 02.04.2021 07:30




Mathematics, 02.04.2021 07:30

Mathematics, 02.04.2021 07:30


English, 02.04.2021 07:30



Chemistry, 02.04.2021 07:30

Chemistry, 02.04.2021 07:30

English, 02.04.2021 07:30

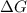 is -618 J/mol
is -618 J/mol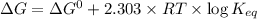
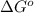 = standard free energy change = +29 kJ/mol =
= standard free energy change = +29 kJ/mol = 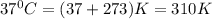
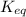 = equilibrium constant =
= equilibrium constant =