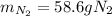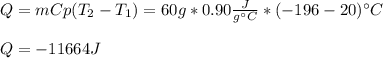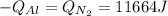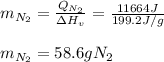Chemistry, 07.06.2020 04:58 isabella4141
A 60 g piece of aluminum at 20°C is cooled to -196°C by placing it in a large container of liquid nitrogen at that temperature. How much nitrogen is vaporized? (Assume that the specific heat of aluminum is constant and is equal to 0.90 kJ/kg·K and that the vaporized nitrogen's temperature does not change.)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2
You know the right answer?
A 60 g piece of aluminum at 20°C is cooled to -196°C by placing it in a large container of liquid ni...
Questions



Advanced Placement (AP), 02.04.2020 21:27




Mathematics, 02.04.2020 21:27


Mathematics, 02.04.2020 21:27

Mathematics, 02.04.2020 21:27


Mathematics, 02.04.2020 21:27


Mathematics, 02.04.2020 21:27


Mathematics, 02.04.2020 21:27

Mathematics, 02.04.2020 21:27

English, 02.04.2020 21:27