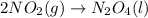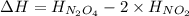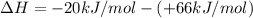Chemistry, 07.06.2020 13:57 Spence8900
PLZ HELP For the reaction: 2NO2(g) → N2O4(l),
the ΔH of the reactants, two moles of NO2 (g), is + 66 kJ/mol,
and the ΔH of the products, N2O4 (l), is -20 kJ/mol.
Which of the following shows the ΔH (change in enthalpy) for the reaction as a whole?
Question 8 options:
ΔrxnH =(- 20 kJ/mol) / (+66 kJ/mol)
ΔrxnH = (+66 kJ/mol) + (- 20 kJ/mol)
ΔrxnH =(-20 kJ/mol) - (+ 66 kJ/mol)
ΔrxnH =(+66 kJ/mol) - (- 20 kJ/mol)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1
You know the right answer?
PLZ HELP For the reaction: 2NO2(g) → N2O4(l),
the ΔH of the reactants, two moles of NO2 (g), is + 6...
Questions

Mathematics, 05.03.2021 05:00

Mathematics, 05.03.2021 05:00

Mathematics, 05.03.2021 05:00

Chemistry, 05.03.2021 05:00

Mathematics, 05.03.2021 05:00

Mathematics, 05.03.2021 05:00


Mathematics, 05.03.2021 05:00


Mathematics, 05.03.2021 05:00

Mathematics, 05.03.2021 05:00





History, 05.03.2021 05:00




Arts, 05.03.2021 05:00

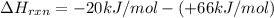
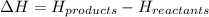
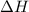 = enthalpy change = ?
= enthalpy change = ?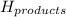 = enthalpy of products
= enthalpy of products 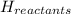 = enthalpy of reactants
= enthalpy of reactants