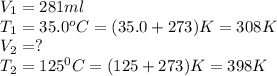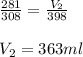Chemistry, 08.06.2020 09:57 brucewayne8499
Carbon dioxide gas at a temperature of 35.0 C and a volume of 281mL. Determine the volume of the gas when the temperature of the gas is raised to 125 C if the pressure remains constant.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2


Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1
You know the right answer?
Carbon dioxide gas at a temperature of 35.0 C and a volume of 281mL. Determine the volume of the gas...
Questions



Engineering, 26.10.2020 17:10



Chemistry, 26.10.2020 17:10

English, 26.10.2020 17:10


Mathematics, 26.10.2020 17:10


English, 26.10.2020 17:10









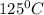 is 363 ml
is 363 ml
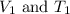 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.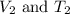 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.